On the Trails: Wringing information out of wood
Published 10:30 pm Sunday, February 20, 2022



By Mary F. Willson
For the Juneau Empire
After some big winds, there were lots of downed trees over the trails. Trail crews soon cleared the ways, leaving the cut ends of tree trunks where we could count the growth rings, if we chose to do so. Seeing those exposed rings, plus thinking about the twisted trunks of some pines, made me contemplate wood a bit — wood as a biological entity, not as a commodity to be sold or a nuisance to be removed.
Growth rings are the cross-sections of xylem tissue (as botanists call it, from the Greek work for wood), constituting what we call wood. In temperate zones, rings are produced annually by living trees. Underneath the bark is a special tissue called cambium, which lays down xylem cells toward the center of the tree and other tissues, called phloem, on the bark side. Phloem carries carbohydrates synthesized by the leaves down to other parts of the tree; special horizontally oriented ray cells bring those carbos from the phloem to the xylem. The xylem tissues provide water and mineral transport from the roots to the leaves, support, and water storage. There is thought to be a limit (set by gravity and other factors) to how high a tree can lift a column of water (between 400 and 426 feet), and some redwoods come close.
In general, wide growth rings indicate good years for growth and narrow ones indicate poor years. The first wood laid down in spring has larger, thinner-walled cells, so it is less dense and light in color, while darker, denser wood is added later in the season. This color contrast is what makes it relatively easy to count the rings. Sometimes the rings are not symmetrical—wider on one side of the tree than the other. A tree that leans (e.g., if exposed to regular strong wind pressure or a fallen one presses against it) can develop more wood on the side away from the pressure, helping to make the tree grow upright.
Each layer of xylem contains a variety of cell types, including starch storage cells and others. But here I want to focus on the cells that comprise the hydraulic, water-conducting system. The passage of water in this pipeline is controlled partly by the amount of water taken in by the roots, partly by conditions along the pipeline, and mainly by evaporation from leaves at the upper end (called transpiration by botanists). Water molecules are polar (with a positive and a negative side), so they tend to cling together, and the entire column of water is pulled up when these molecules move from the leaves to the air. The pipeline is very narrow, and capillary action with the sides of the pipe makes water movement relatively easy. Leaves have openings called stomata that can open to keep water moving or close to conserve water. This system can run into trouble if the tree is subjected to freeze-thaw cycles when frozen fluid pushes out air bubbles that tend to break the water column and stop the flow (an embolism, as sometimes form in animals’ blood vessels). The problem can be exacerbated if the water supply is low, as during a drought.
As a tree grows and ages, the older xylem stops conducting water and becomes darker; it retains its support and storage functions (we call it ‘heartwood’). The younger wood (‘sapwood’) has the water-conducting function but is also important structural support.
Although wood density may be mostly genetically controlled, growing conditions (shadiness, soil fertility and moisture, etc.) can also influence characteristics of the hydraulic system, including the amount of leaf surface and stomata that release water and the water-conducting cells themselves. Most tree populations exhibit considerable genetic and environmentally plastic variation.
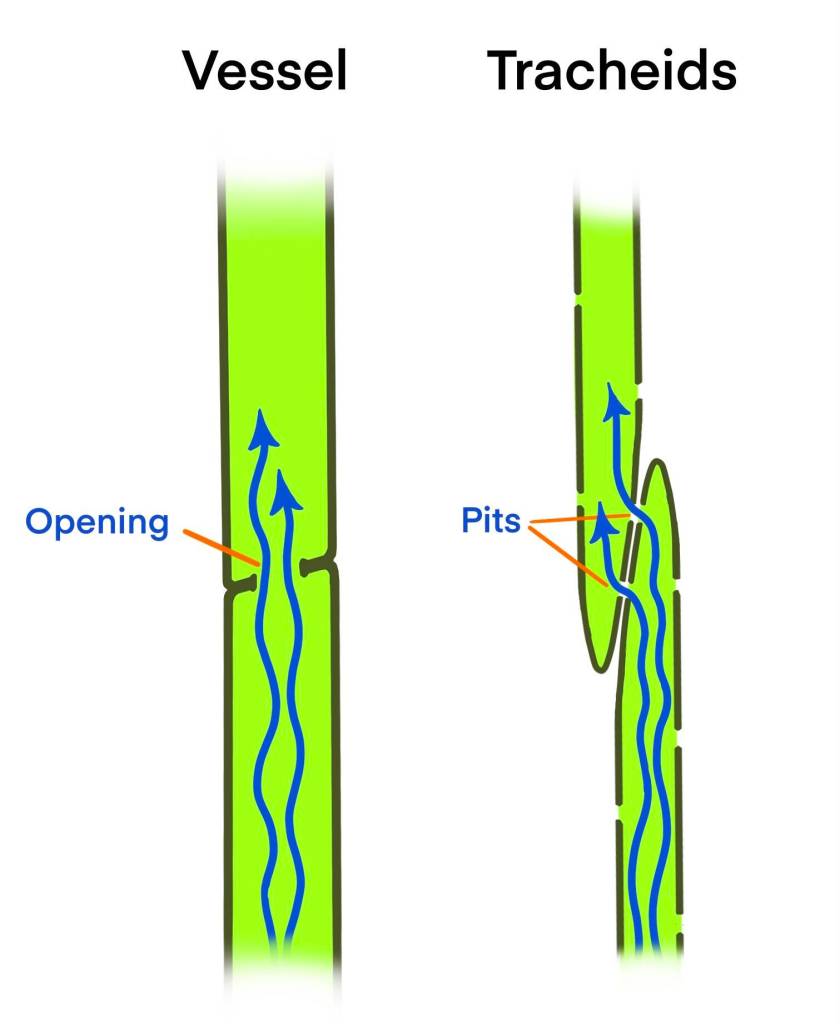
The water-conducting pipelines of conifers and flowering plants (called angiosperms—woody species include maples, alders, oaks, etc.) are different. Conifer pipelines are composed of ‘tracheids’, spindle-shaped cells, tapered at both ends, with lots of pits along the sides. Adjacent tracheids are offset from each other but their pits are matched up, so water can zigzag from one to another. In a tall tree, water may pass through many thousands of tracheids and matched pits on its way up to the leaves. These pits have an intricate, specialized valve that controls flow from one cell to the next, contributing to a tracheid’s ability to recover from an embolism and reducing the risk of an embolism passing from one tracheid to another. Conifer water-transport systems are relatively narrow and resistant to freeze-thaw cycles. Most of the structural support in conifer xylem comes from the tracheids. These cells die as they age but can maintain their conducting function for years.
The woody angiosperms have some tracheids too, but structural fibers do most of the support and (in most species) their principal water-conducting pipeline is made of cells called ‘vessels’. More columnar and wider than tracheids, these cells have sizable openings at each end, guarded by valve-like control devices. They line up one above another, and water can move straight up the pipe. This arrangement is thought to give these plants greater capacity for water movement, but they seem to be more vulnerable to freeze-thaw cycles than conifers. In one study, comparisons of many angiosperm species found that those with greater transport efficiency had less embolism resistance, and these differences were associated with water availability in the species’ habitats. More comparative studies will surely discover more variations that are related to environmental circumstances.
Details of these hydraulic systems and their functioning are the domain of hydraulic engineers!
• Mary F. Willson is a retired professor of ecology. “On the Trails” appears every Wednesday in the Juneau Empire.